https://www.youtube.com/c/chemigod.
Practical Scheme 2023-24
| Part | Marks in CBSE India | Category | Name of Experiment | Remark |
| A | 8 | Volumetric Analysis | Titration of potassium permanganate with Mohr,s salt | |
| A | Titration of potassium permanganate with oxalic acid | |||
| B | 8 | Salt Analysis | (Given 11 Chemistry Tab)-This Portion is common in both 11 and 12 standard | |
| C | 6 | Content Based Experiments | A. Surface Chemistry (a) Preparation of one lyophilic and one lyophobic sol Lyophilic sol – starch, egg albumin and gum Lyophobic sol – aluminium hydroxide, ferric hydroxide, arsenous sulphide. (b) Dialysis of sol-prepared in (a) above. (c) Study of the role of emulsifying agents in stabilizing the emulsion of different oils. B. Chemical Kinetics (a) Effect of concentration and temperature on the rate of reaction between Sodium Thiosulphate and Hydrochloric acid. (b) Study of reaction rates of any one of the following: (i) Reaction of Iodide ion with Hydrogen Peroxide at room temperature using different concentration of Iodide ions. (ii) Reaction between Potassium Iodate, (KIO3) and Sodium Sulphite: (Na2SO3) using starch solution as indicator (clock reaction). C. Thermochemistry Any one of the following experiments i) Enthalpy of dissolution of Copper Sulphate or Potassium Nitrate. ii) Enthalpy of neutralization of strong acid (HCI) and strong base (NaOH). iii) Determination of enthaply change during interaction (Hydrogen bond formation) between Acetone and Chloroform. D. Electrochemistry Variation of cell potential in Zn/Zn2+|| Cu2+/Cu with change in concentration of electrolytes (CuSO4 or ZnSO4) at room temperature. E. Chromatography i) Separation of pigments from extracts of leaves and flowers by paper chromatography and determination of Rf values. ii) Separation of constituents present in an inorganic mixture containing two cations only (constituents having large difference in Rf values to be provided). F. Preparation of Inorganic Compounds Preparation of double salt of Ferrous Ammonium Sulphate or Potash Alum. Preparation of Potassium Ferric Oxalate. G. Preparation of Organic Compounds Preparation of any one of the following compounds i) Acetanilide ii) Di -benzalAcetone iii) p-Nitroacetanilide iv) Aniline yellow or 2 – Naphthol Anilinedye. H. Tests for the functional groups present in organic compounds: Unsaturation, alcoholic, phenolic, aldehydic, ketonic, carboxylic and amino (Primary) groups. I. Characteristic tests of carbohydrates, fats and proteins in pure samples and their detection in given foodstuffs. | |
| D | 4 + 4 | 4 Project Work and Viva (Internal and External Both) | You have to prepare your self while performing experiments | |
| Total Marks | 30 | – | – | – |
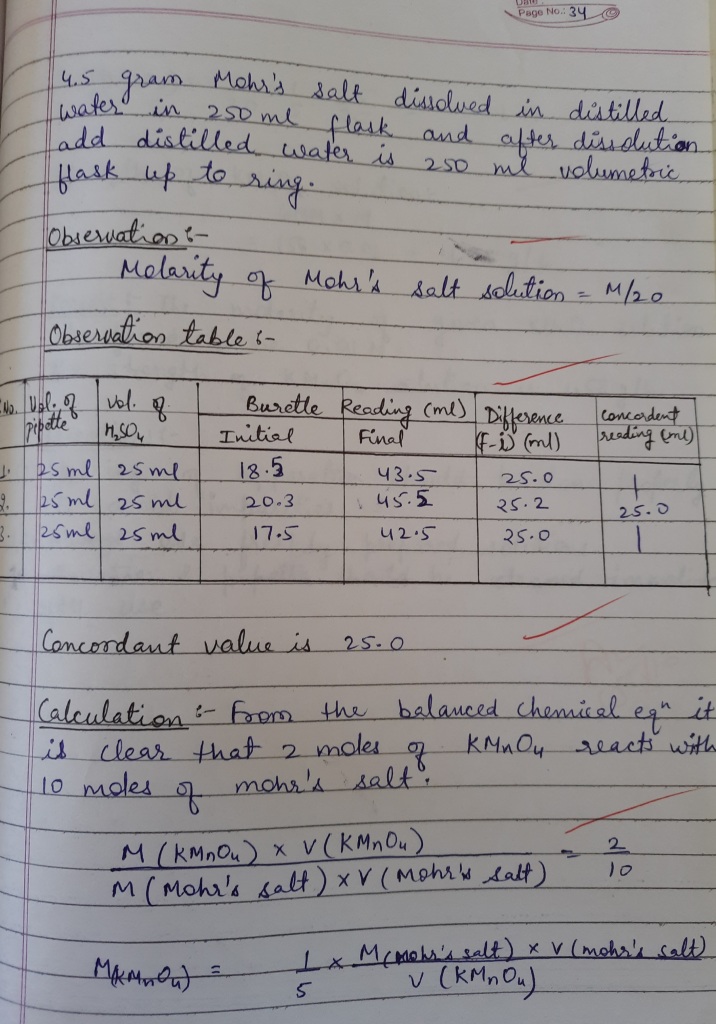
Titration of potassium permangnate with Mohr,s salt


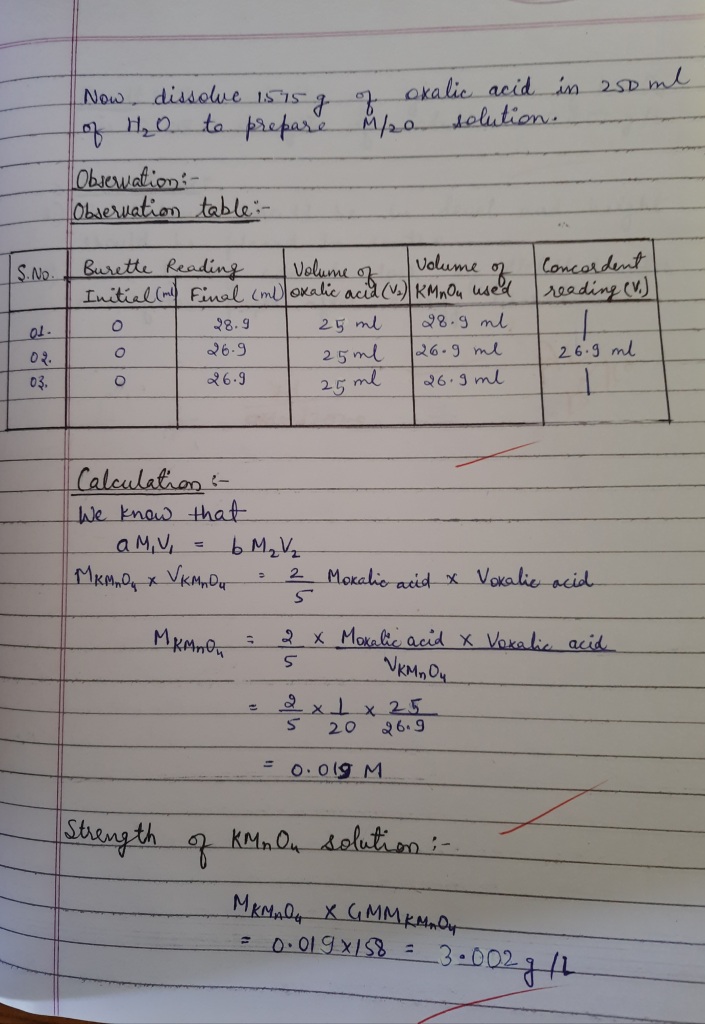
Titration of potassium permanganate with oxalic acid- ChemiGod


Content based Exp – Distinguish test-Acetic acid and Ethanol- ChemiGod

Content based Exp – Distinguish test-Aniline and phenol


